Crystallography: From the arrangement of atoms to a world of possibilities
Tania Firouzabady. 03/12/2021

Though it is common knowledge that matter is composed of atoms, the way atoms are organized is often overlooked. Crystallography is a branch of science dealing with the arrangement of atoms in crystalline solids.
Crystallographers determine how atoms are organized by uncovering the properties and structures of the crystals through various techniques, such as X-rays and electron diffraction, the technique of firing electrons at a sample of matter to observe the interference pattern. These properties include the chemical makeup of solids and reveal how they would react to external forces like temperature and salinity. This information is then used by scientists in other fields, including chemistry, biology and physics.
On a smaller scale, a primary aspect of crystallography is the study of the crystal lattice. A lattice site can be thought of as a representative point; since atoms are arranged symmetrically and at regular intervals, each group of atoms can be represented as a single point. Hence, the entire crystal lattice is a structured arrangement of infinite points with the lattices comprising the outline of the repeating patterns.
Geometrically, the arrangement of a crystal lattice is characterized by the intersection of three parallel lines. If you were to separate the lines, there would be three distinct planes—arranging them in a particular manner will result in a 3D network resembling a cube. Each cube is a vital component of the symmetry of a crystal lattice.
A common method used by crystallographers is X-ray crystallography. Through this technique, an X-ray beam is diffracted into different directions when it passes through crystalline atoms. Crystallographers measure the angles of the beams to create a three-dimensional model of the electrons’ densities. Electron density is valuable as it allows them to analyze the positions of the atoms, locations of chemical bonds and more. Although X-ray crystallography provides a vast amount of information, it also has shortcomings: the samples of crystals typically need to be placed in an environment that can cause conformational changes or changes to the structure of the crystal. Furthermore, certain crystals are hard to analyze using this method, such as virus particles. Nonetheless, it offers many advantages and has been used extensively in the past.
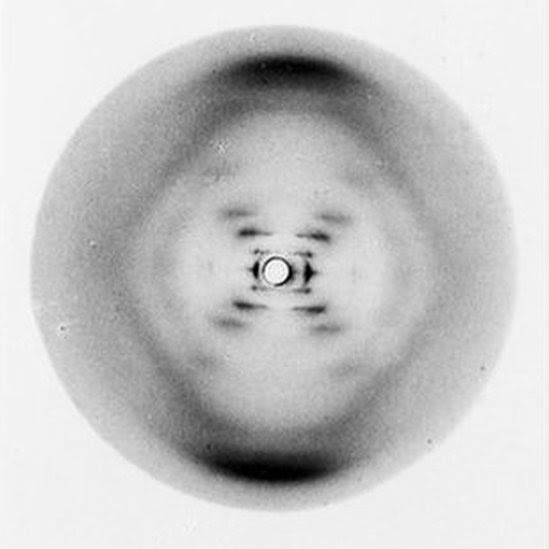
A notable example of X-ray crystallography is the role it played in the discovery of DNA’s double helix. On May 6, 1952, Rosalind Franklin discovered the three-dimensional structure of DNA, which led to the model created by Watson and Crick. According to The Embryo Project Encyclopedia, “Photograph 51, or Photo 51, revealed information about DNA’s three-dimensional structure by displaying the way a beam of X-rays scattered off a pure fiber of DNA.” As previously mentioned, the diffraction of the beams provides information about a material’s structure. The publication also states, “some biological macromolecules, such as DNA, can form fibers suitable for analysis using X-ray crystallography because their solid forms consist of atoms arranged in a regular pattern.” In other words, not all macromolecules can be analyzed with X-ray crystallography—only those with a regular atom arrangement. This proves to be the case for DNA.
More recent applications of crystallography include its use in the pharmaceutical industry. X-ray crystallography has served as a key source of data for the development of new drugs. As outlined by the National Center for Biotechnology Information (NCBI), “The remarkable progress in methods for recombinant protein production and crystallization in recent years is in part due to the success of various protein structure and function initiatives.” Protein crystallization is the process of stabilizing protein molecules by crystal contacts. This ultimately allows scientists to see details, such as mutations, in the proteins that are not detectable by even the most powerful light microscopes. The NCBI further describes how recent technological advancements have allowed researchers to apply crystallization more efficiently and have “improv[ed] the probability of crystallization of macromolecules.”
A specific example of X-ray crystallography being used for a major development is with regards to insulin, a hormone that regulates blood glucose levels. Since 1926, scientists have been searching for a way to obtain photographs of the crystals in insulin, as described by Dorothy Crowfoot in an article published in Nature. The first attempts made by W.H. George were unsuccessful. However, later studies revealed that pepsin, a digestive enzyme, “could give a single X-ray diffraction pattern, suggesting that insulin… could be attacked in the same way if large enough crystals could be grown.” Studies of insulin’s crystallization by D.A. Scott made this possible. By examining its crystal structure, scientists are better able to address some of the problems regarding insulin—too much or too little insulin in the bloodstream interferes with normal glucose levels and is associated with diabetes.
Currently, scientists are using crystallography to study the SARS-CoV-2 Coronavirus that is responsible for COVID-19. They have already discovered pieces (such as protein molecules) that make up the virus. Evidently, crystallography holds great potential for future research. Understanding the arrangement of atoms within crystalline solids allows for advancements to be made in a variety of scientific fields.
Cover Photo: (The Scientist)
